|
3/10/2024 0 Comments Bohr model of nitrogen atom
When drawing a Bohr diagram, the valence electrons would be present in the outermost electronic level/shell (furthest away from the nucleus). Valence electrons are located in the highest energy level of an atom. In atomic physics, the Bohr model or RutherfordBohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons. Bohr model of sulfur: (CC BY-SA 2.0 uk Greg Robson). This simulation is part of the PhET project, a leading provider of free online STEM resources. Here’s how you can draw the Bohr model of nitrogen step by step. You can also play a fun game to check your understanding of atomic concepts. Watch this video of your professor drawing Bohr models and identifying valence electrons. Bohr model of calcium: (CC BY-SA 2.0 uk Greg Robson): Answer b. Do you want to learn how to build an atom from scratch Try this interactive simulation and explore the structure and symbols of atoms, isotopes, and ions. 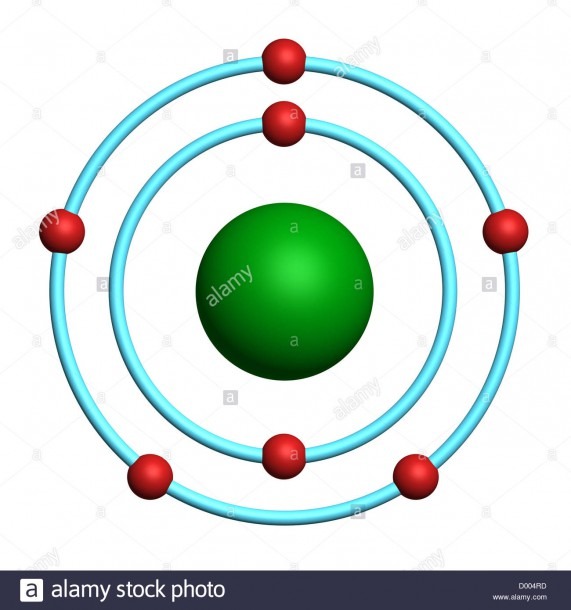
This process will allow us to identify the valence or active electrons of these particular elements. 
We will also be limited in drawing Bohr diagrams of only the first twenty elements on the periodic table. In other words, we will limit each shell after n=1 to a maximum of eight total electrons. In this course, we will be using a modified Bohr model. The orbits are labeled by an integer, the quantum number n. Number of neutrons can be calculated using the formula, Number of neutrons = mass number – number of protons.\): Bohr models of different groups/families of elements from the periodic table. In the Bohr model of the atom, electrons travel in defined circular orbits around the nucleus. In the year 1913, Niels Bohr proposed an atomic structure model, describing an atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the positively charged nucleus like planets around the sun in our solar system, with attraction provided by. Bohr model of calcium: (CC BY-SA 2.0 uk Greg Robson): Answer b. Rutherford’s model introduced the nuclear model of an atom, in which he explained that a nucleus (positively charged) is surrounded by negatively charged electrons. Bohr model of nitrogen: (CC BY-SA 2.0 uk Greg Robson) Answer b. It came into existence with the modification of Rutherford’s model of an atom. Now, since the number of protons and electrons are always equal in any atom, the total electrons in nitrogen is 7. Bohrs model of atom: The Bohr model of the atom was proposed by Neil Bohr in 1915. This means that there are a total of 7 protons in the nitrogen atom because the atomic number is nothing but the total number of protons in it. Now, in the above given atom that is nitrogen $N$, After the first shell, the next shell is drawn and this is a continuous process where it is different for every different atom. Next, the first outer shell is drawn in which according to the electronic configuration rule, only two electrons can be accommodated. It just takes a little hard work and a basic understanding of an atom. A Bohr model of a chlorine atom shows a nucleus surrounded by three concentric rings. Check how the prediction of the model matches the experimental results. The first shell of the Bohr diagram of Nitrogen has 2 electrons, and the second shell has 5 electrons. The Bohr model of the atom was proposed by Neil Bohr in 1915. This nucleus is surrounded by two electron shells. Bohr’s atomic model consists of the central nucleus which is drawn as the circle and then the total number of protons and neutrons are written accordingly. Try out different models by shooting light at the atom. The Bohr Model of Nitrogen (N) has a nucleus with 7 neutrons and 7 protons. Let us now focus on the Bohr’s atomic model and how it is represented. In the classes of chemistry, we have come across the concepts of atoms and also some the definitions relating to it such as the atomic number, atomic mass, composition of atom, properties of the atom etc. Hint: The answer here is based on the calculation of number of protons, neutrons and the electrons and then drawing the Bohr’s model accordingly whether the nucleus has protons and neutrons accommodated in it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
